Position-specific isotope analysis (PSIA) is a technique that measures the isotope composition of a certain position in a molecule (such as the δ13C value of methyl group in acetic acid). It can provide valuable information of the formation mechanisms of a molecule.
Butane (C4H10) is one component of natural gas, an atmospheric organic pollution on Earth, and an important compound in chemical and petrochemical industries, which also represents a typical hydrocarbon with an abiotic origin under deep Earth conditions, and an astrophysical molecule on extraterrestrial planets and their satellites. It has two structural isomers: normal butane (n-butane) and isobutane (i-butane). What shall the position-specific isotope effects in butane tell us?
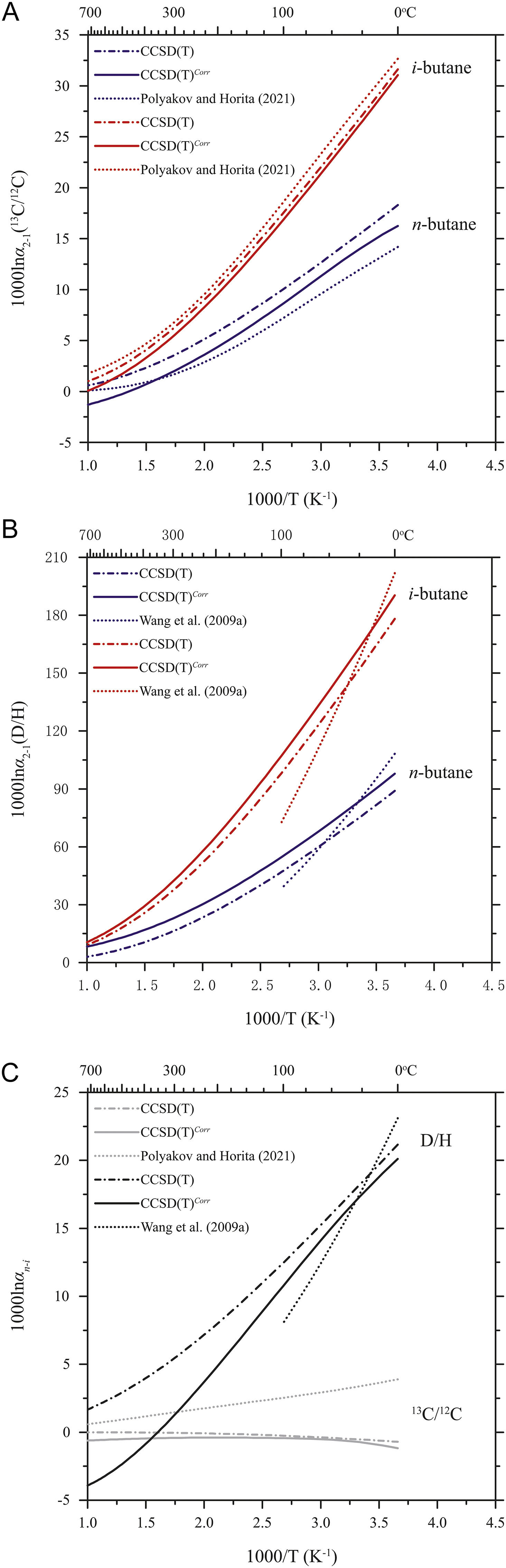
An international research group led by Prof. LIU Yun from the Institute of Geochemistry of the Chinese Academy of Sciences (IGCAS) conducted quantum chemistry modeling with 8 corrections (such as anharmonic correction, hindered internal rotation correction, and diagonal Born-Oppenheimer correction) beyond the harmonic approximation and the Born-Oppenheimer approximation to obtain accurate intramolecular and intermolecular carbon and hydrogen isotope fractionation factors for butane isomers at equilibrium.
The researchers also calculated the abundance ratio of n-butane to i-butane at equilibriums at various temperatures. They found that the previous calculations of isotope fractionations using "cutoff" treatment were not suitable for the study of isotope effects in and between butane isomers.
Meanwhile, their results showed that the equilibrium position-specific isotope fractionation factor of i-butane was almost twice the value of n-butane for either carbon or hydrogen isotope substitutions, which might serve as an indicator to identify the processes related to the isomerization of alkanes.
“Our calculation shows that the abundance of i-butane at equilibrium theoretically should be much greater than that of n-butane. However, the calculation contrasts with the observations in natural settings, indicating that the proportion of butane isomers may be controlled by precursor hydrocarbons and formation mechanisms.” said Dr. LIU Qi from IGCAS, “For example, the proportion may inherit from kerogen and be modified by kinetic processes during formation, and then the cracking processes of butane may also affect the isotope distributions in methane, ethane, and propane.”
Their calculation results can be further utilized for calibrating experimental measurements, establishing new geothermometers, and recognizing kinetic isotope effects.
The researchers prospect the potential application of intramolecular isotope equilibriums as a geothermometer as well as a tracer for microbial oxidations and abiotic origins of hydrocarbons.
The study was published in Chemical Geology on Dec. 17. (Liu Q., Yin X., Zhang Y., Julien M, Zhang N., Gilbert A., Yoshida N., Liu Y.* (2021) Theoretical calculation of position-specific carbon and hydrogen isotope equilibriums in butane isomers. Chemical Geology 561: 120031. )
|
|
|
Equilibrium isotope fractionation factors (1000lnα) within and between butane isomers compared with the previous studies on the harmonic approximation (Image by IGCAS) |
Contacts:LIU Qi; LIU Yun
Institute of Geochemistry, Chinese Academy of Sciences
Email: liuqi@mail.gyig.ac.cn; liuyun@mail.gyig.ac.cn
(By Prof. LIU Yun’s group)